Meeting Calendar
Events in April 2024
MMonday TTuesday WWednesday TThursday FFriday SSaturday SSunday 11-Apr-202422-Apr-202433-Apr-202444-Apr-202455-Apr-202466-Apr-202477-Apr-202488-Apr-202499-Apr-20241010-Apr-20241111-Apr-20241212-Apr-20241313-Apr-20241414-Apr-20241515-Apr-20241616-Apr-20241717-Apr-20241818-Apr-20241919-Apr-20242020-Apr-20242121-Apr-20242222-Apr-20242323-Apr-20242424-Apr-20242525-Apr-20242626-Apr-20242727-Apr-20242828-Apr-20242929-Apr-20243030-Apr-202411-May-202422-May-202433-May-202444-May-202455-May-2024-
Recent Posts
Recent Comments
- M. Rogers on Latest Draft of EDM Sub Reference Model
- Thierry Felix on Latest Draft of EDM Sub Reference Model
- Silvio Mann on Latest Draft of EDM Sub Reference Model
- Carl Sampson on Latest Draft of EDM Sub Reference Model
- Romuald on Device Reference Model Team Formed
Archives
Meta
Author Archives: David Gwyn
Announcing the DIA EDM Structured Submission Reference Model
We have come a long way since the first version of the EDM Submission Reference Model was drafted in 2009. With an eye on the future, the model was designed to be independent of any single software solution and to … Continue reading
Posted in Submission Ref Model
Comments Off on Announcing the DIA EDM Structured Submission Reference Model
Latest Draft of EDM Sub Reference Model
The latest draft of the EDM Submission Reference Model is available for review. This includes the latest content regarding prescribing information and also updates to the Regulatory / Administrative information. It can be downloaded at the link below: https://edmrefmodel.com/download/edm-submission-reference-model-20-draft/
Posted in Submission Ref Model
9 Comments
FW: TMF RM Tech Committee – Presentation Subgroup Kick-off
Meeting Date: 2/12/2015 12:00 PM Location: GoToMeeting A – Corporate (Paul) Notes The goals of the team were discussed and augmented during the meeting. The goals identified are: Define alternative forms for consuming the data produced by the … Continue reading
Posted in TMF RM Tech Presentation
Comments Off on FW: TMF RM Tech Committee – Presentation Subgroup Kick-off
Integration with TMF Reference Model
Today’s agenda for the EDM Submission Reference Model team includes a discussion on the overlap and integration between the EDM Submission and TMF Reference models. While the TMF model was spawned from the EDM team a number of years ago, … Continue reading
Posted in Submission Ref Model
Comments Off on Integration with TMF Reference Model
Device Reference Model Team Formed
We are excited to announce that a new subcommittee has formed to support development of a reference model for medical device submissions. The first meeting of the subcommittee is scheduled on Wednesday, January 14th at 12:00 – 1:00 EST. Please … Continue reading
Posted in Device Ref Model
1 Comment
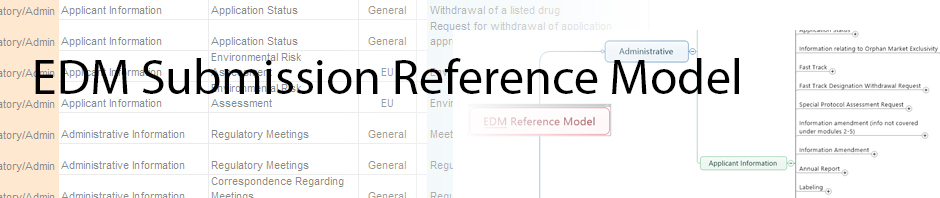
Mind Map of Reference Model
In an effort to modernize our delivery of reference model information, the team used techniques that we use in content management projects which include mind mapping techniques for visualization. We have created a [wpdm_hotlink id=’88’ link_label=’mind map’] of the model … Continue reading
Posted in Submission Ref Model
Comments Off on Mind Map of Reference Model